china tio2 p25
Titanium dioxide, a naturally occurring compound, is widely used in various industries due to its unique properties. This versatile white pigment is known for its excellent whiteness, opacity, and chemical stability. In this article, we will discuss the benefits of wholesale titanium dioxide and explore its applications in different sectors.
Titanium dioxide (TiO2) is by far the most suited white pigment to obtain whiteness and hiding power in coatings, inks and plastics. This is because it has an extremely high refractive index and it does not absorb visible light. TiO2 is also readily available as particles with the right size (d ≈ 280 nm) and the right shape (more or less spherical) as well as with a variety of post-treatments.
However, the pigment is expensive, especially when the volume prices of systems are used. And, there always remains a need to develop a full-proof strategy to obtain the best results in terms of cost/performance ratio, scattering efficiency, dispersion… while using it in coating formulations. Are you searching for the same?
Explore the detailed knowledge of TiO2 pigment, its scattering efficiency, optimization, selection, etc. to achieve the best possible white color strength and hiding power in your formulations.
However, the pigment is expensive, especially when the volume prices of systems are used. And, there always remains a need to develop a full-proof strategy to obtain the best results in terms of cost/performance ratio, scattering efficiency, dispersion… while using it in coating formulations. Are you searching for the same?
Explore the detailed knowledge of TiO2 pigment, its scattering efficiency, optimization, selection, etc. to achieve the best possible white color strength and hiding power in your formulations.
 When combined with other materials, TiO2 forms a photoactive layer that absorbs sunlight and generates electricity When combined with other materials, TiO2 forms a photoactive layer that absorbs sunlight and generates electricity
When combined with other materials, TiO2 forms a photoactive layer that absorbs sunlight and generates electricity When combined with other materials, TiO2 forms a photoactive layer that absorbs sunlight and generates electricity




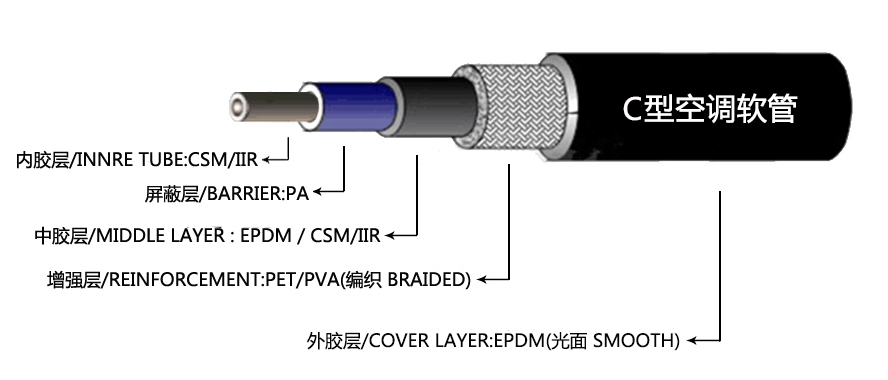 Over time, exposure to heat, cold, and road elements can cause hoses to deteriorate, potentially causing cracks or bulges Over time, exposure to heat, cold, and road elements can cause hoses to deteriorate, potentially causing cracks or bulges
Over time, exposure to heat, cold, and road elements can cause hoses to deteriorate, potentially causing cracks or bulges Over time, exposure to heat, cold, and road elements can cause hoses to deteriorate, potentially causing cracks or bulges
 Their compact size enables them to navigate tight spaces and reach areas that would be inaccessible to larger equipment Their compact size enables them to navigate tight spaces and reach areas that would be inaccessible to larger equipment
Their compact size enables them to navigate tight spaces and reach areas that would be inaccessible to larger equipment Their compact size enables them to navigate tight spaces and reach areas that would be inaccessible to larger equipment These innovations integrate seamlessly with the traditional mechanical functions, enhancing the driving experience and providing drivers with added peace of mind These innovations integrate seamlessly with the traditional mechanical functions, enhancing the driving experience and providing drivers with added peace of mind
These innovations integrate seamlessly with the traditional mechanical functions, enhancing the driving experience and providing drivers with added peace of mind These innovations integrate seamlessly with the traditional mechanical functions, enhancing the driving experience and providing drivers with added peace of mind